Enhancing the Electrocatalytic Activity of Molybdenum Disulfide
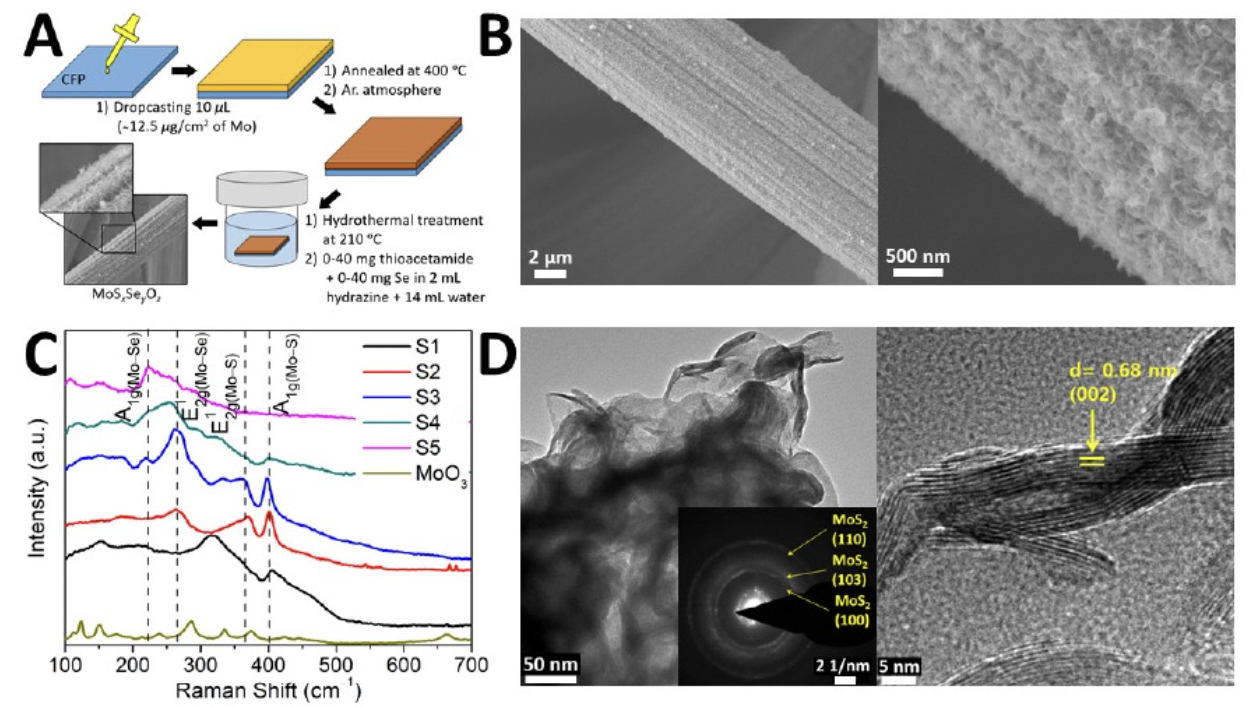
A) Illustration of the step-wise synthetic process comprising drop-casting, thermal annealing, and hydrothermal sulfidation/selenization used to grow MoS2-xSex/MoO3 nanosheets on carbon fiber paper. (B) SEM image of sample where Se:S=0.48 showing the homogeneous distribution of the nanosheets on carbon filter paper. (C) Raman spectra (514.5 nm excitation) acquired for MoS2-xSex/MoO3 samples with increasing concentration of Se. (D) Low-magnification and HRTEM images of a sample with a Se:S ratio of 0.62 illustrating the layered structure of MoS2/MoSe2. The left inset shows a SAED pattern of the chalcogenide layers.
Sarbajit Banerjee